Some thoughts on Hantavirus
More questions than answers at the moment.
Note: the contents of this post reflect what I was able to find about the situation as of circa 1 AM EST on May 6, 2026. The situation is evolving and this post is already out of date with regard to the status of the situation. I will be putting out more materials as substantive findings emerge, but this post will not be updated further with regard to the epidemiological situation of the MV Hondius hantavirus outbreak.
Bottom line up front:
The probability of MV Hondius’s hantavirus outbreak resulting in a pandemic is nonzero, but extremely low. It is, however, something that warrants close monitoring by experts. There is no call presently for members of the general public to modify their approach to infection prevention beyond what is already recommended.
The specific type of hantavirus has not yet been identified, but based on the location, Andes virus is likely.
There is evidence that Andes virus is capable of human-to-human transmission under conditions of prolonged, close contact and while the infected individual is symptomatic. This is being seriously considered as a means by which the virus spread between passengers (and should be). Nonetheless, a shared environmental exposure is also very much possible.
Andes virus does not transmit efficiently from person-to-person, though it is possible.
The incident has key relevance for the US as it is about to host the World Cup, wherein millions of travelers from abroad will be visiting and may be incubating pathogens during their travels, and may seek care in the US. Having a robust public health infrastructure primed to address potential outbreaks is key in that situation, but under the current administration, our capacity to respond to infectious diseases has been thoroughly damaged. It is worth reading this article in full.
We would do well to remember that while the outbreak on MV Hondius poses little risk to the world as a whole, there are real people on the ship who are justifiably terrified and deserve compassion and recognition of their humanity.
Brief Situation Summary
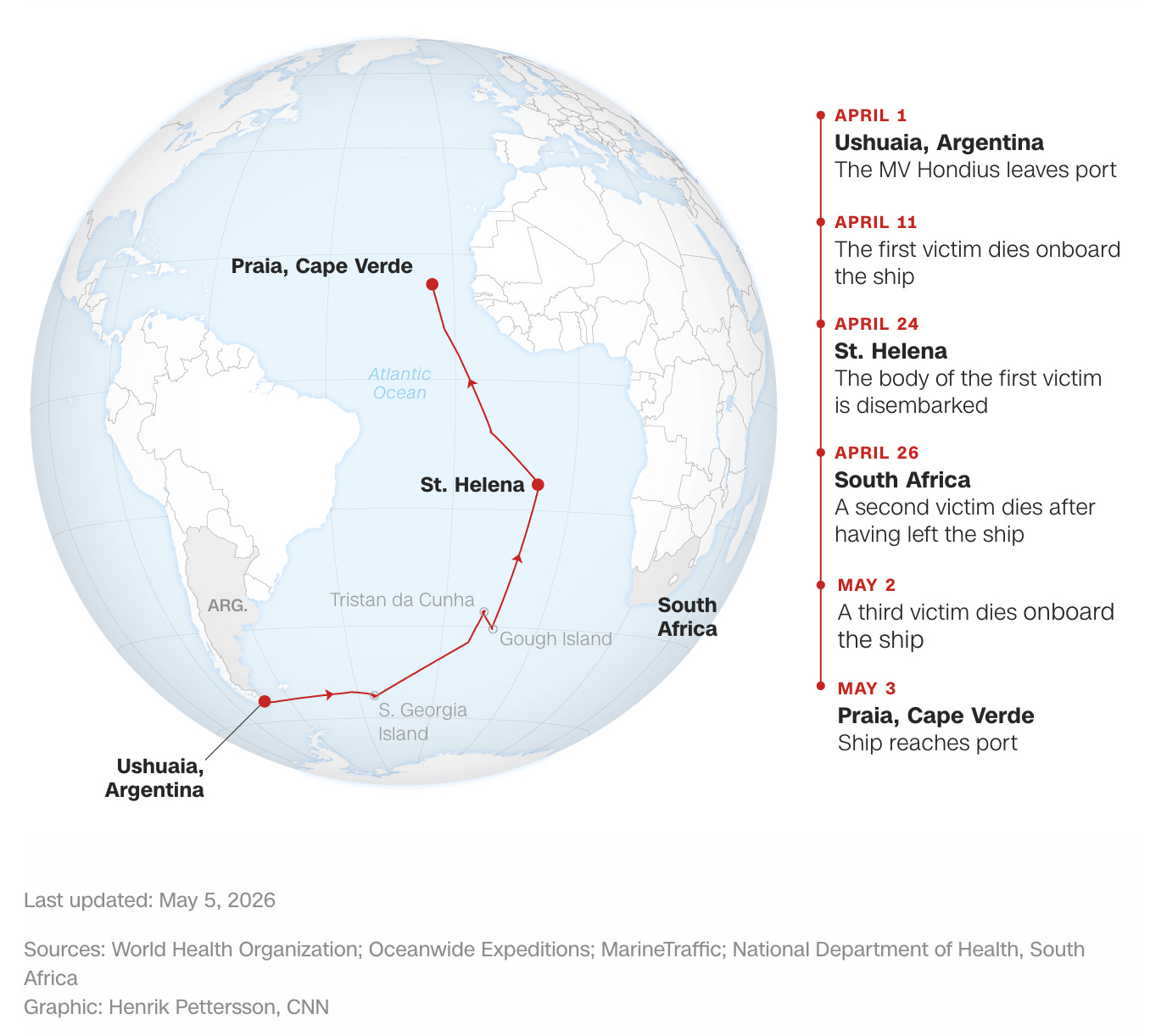
The MV Hondius is a cruise ship that had about 147 individuals (88 passengers and 59 crew) and stopped in Ushuaia, Argentina, on April 1, 2026. On April 11, the first victim died aboard the ship, with deaths from 2 more victims on April 26 and May 2. At the time of writing this, 2 of these cases are confirmed to be hantaviruses, and 5 more are suspected cases. Two of the patients experiencing symptoms right now are crew members. The body of one of the deceased patients is presently on the ship. The first deceased patient was a 70-year-old Dutch man; his body was taken off the vessel in St. Helena and is awaiting repatriation. His wife subsequently began to experience symptoms and was transferred in South Africa, where she collapsed in an airport and died in a hospital. One patient is currently in intensive care in South Africa, and the other suspected cases are experiencing only mild symptoms. A third fatality was reported on May 2, a woman experiencing pneumonia. The ship’s doctor is reported to be in serious condition and will be evacuated in a medical flight to the Canary Islands. Argentina does have reservoir species for transmission of hantavirus, but all current cases this year have been reported in Buenos Aires and none have been reported in the province where the MV Hondius was docked. The WHO suspects that the first case was infected before boarding the ship.
What’s Hantavirus?
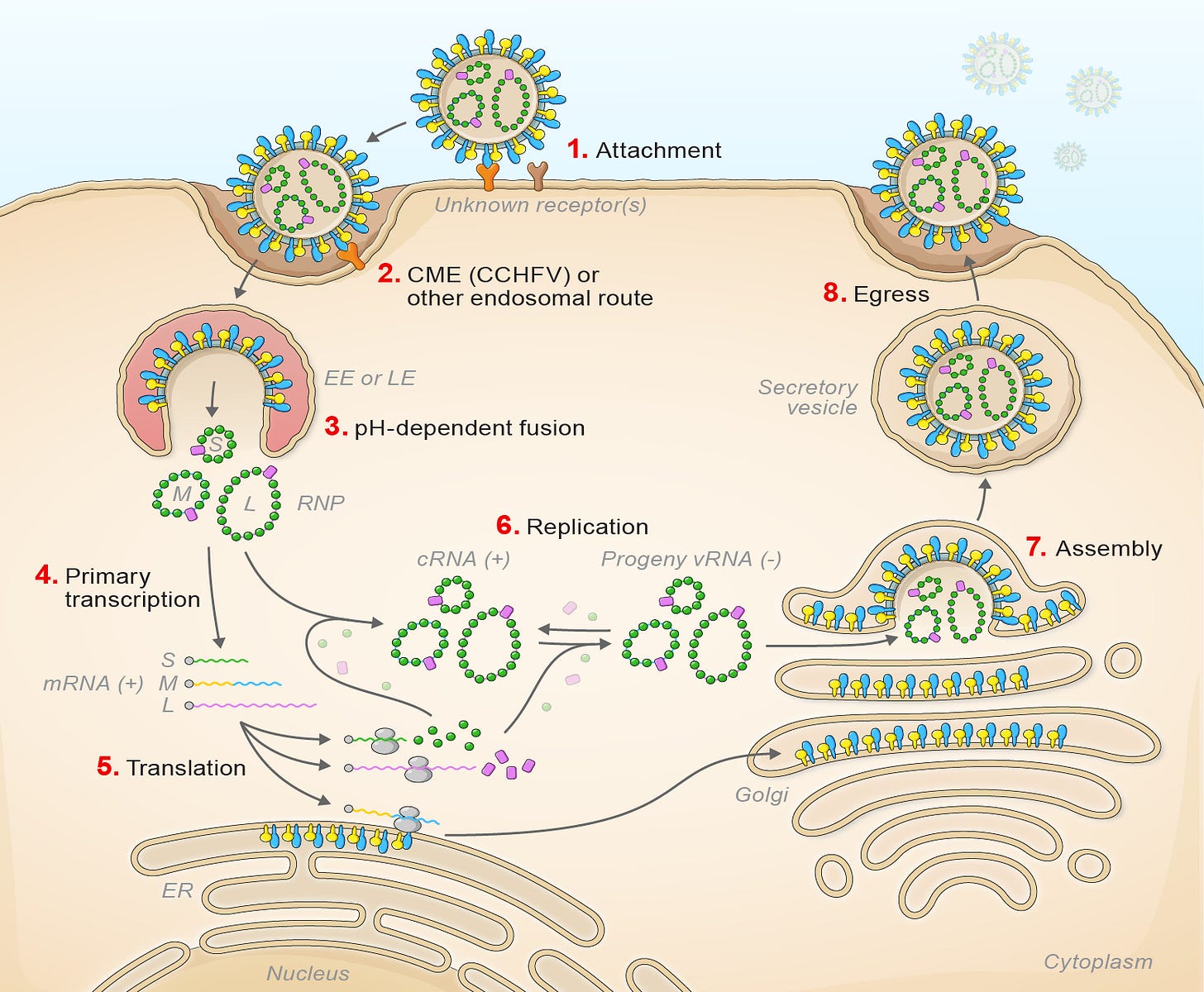
“Hantavirus” is a bit of a confusing term because it does not describe a distinct virus but rather a family of viruses (that is, viruses in the family Hantaviridae), and the orthohantavirus genus is the only one known to cause infections of humans. Hantaviruses have been recognized as one of the 25 viral families with pandemic potential. There is considerable diversity among hantaviruses, and those that infect humans have rodent (usually mice or rats; see Table 1) reservoirs (animals that can be infected without becoming seriously ill and thus can continuously act as a source of infection for others). Hantaviruses are generally contracted through exposure to the urine, feces, or other secretions of infected rodents, which are then inhaled; there is a seasonal pattern seen in infections for certain hantaviruses (e.g., Puumala virus) with cases rising in the winter, suggesting a role for ventilation in transmission. Here’s what the replication cycle looks like for hantaviruses, courtesy of the ICTV:
There are a few important things to note here. First: the viruses are enveloped, single-stranded negative-sense RNA viruses (i.e., the virus’s genome does not specify a protein, but rather has to undergo synthesis of a complementary mRNA strand to generate an mRNA that does). Second, hantaviruses have a segmented genome with 3 linear RNAs (L, M, and S—large, medium, and small) with terminal complementarity that causes them to adopt circular structures. That means that hantaviruses are capable of reassortment—when two (or more) hantaviruses infect the same cell, it is possible to get a hybrid virus which has segments from both viruses as part of its genome. This represents a much faster mode of evolution (when this occurs with influenza viruses, it is very concerning because this is a major path to flu pandemics) than the standard gradual acquisition of mutations through random error (though the viruses are RNA viruses and, unlike coronaviruses, do not have proofreading enzymes to ensure the fidelity of replication). Nonetheless, the comparison is not exact because hantaviruses are more restricted in the species they infect than influenza viruses. This creates a problem in terms of trying to get an effective vaccine against hantaviruses broadly. There are 4 major products encoded by the genomes of Hantaviruses:
Nucleoprotein (N): Encapsulates viral RNA and helps ensure it is packaged into newly assembled particles. Encoded by the S RNA segment.
Glycoprotein (Gn/Gc): Essentially the hantavirus equivalent of spike for coronaviruses or hemagglutinin for influenza viruses. The glycoprotein has 2 subunits, Gn and Gc (N-terminal and C-terminal fragments respectively), which assemble into tetramers a tetramer of Gn subunits in the center and then a tetramer of Gc subunits in the periphery. These proteins facilitate viral entry into cells. Encoded by the M RNA segment.
Large protein (L): Enables replication of hantavirus RNA and engages in cap snatching to (1) enable the production of viral proteins and (2) hide the viral RNA from the cell to avoid tripping an immune response that suppresses replication. Encoded by the L RNA segment.
Nonstructural proteins: These vary in function and across hantaviruses but basically serve to inhibit the induction of the immune response so that hantaviruses have a chance to replicate. They are not incorporated into new viral particles. These are found in the S segment (when present).
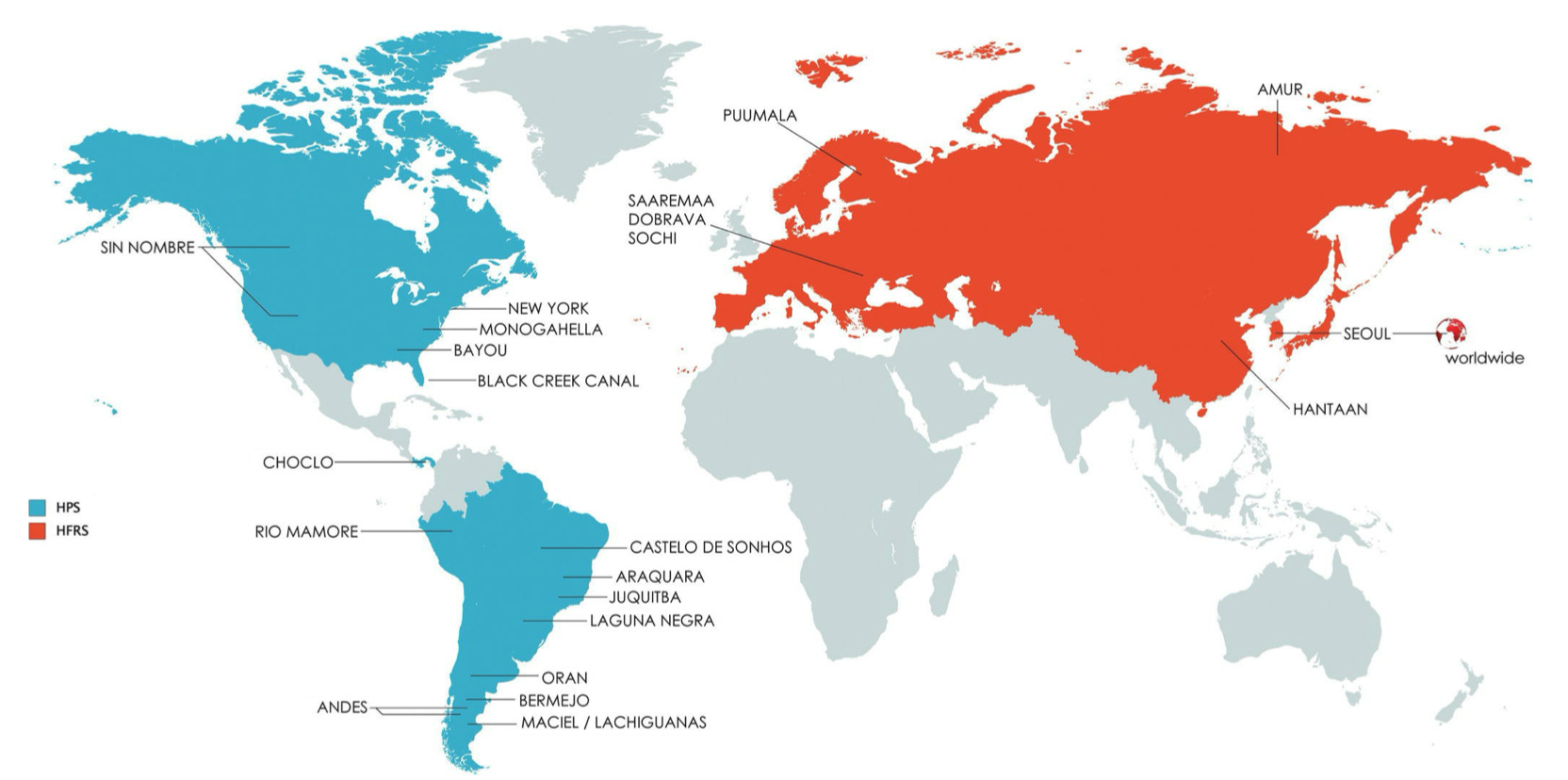
Hantaviruses can further be divided into New World hantaviruses and Old World hantaviruses (see Figure 17.3 from Fields Virology: Emerging Viruses 7th Edition below; New World viruses are in blue and Old World viruses are in red).
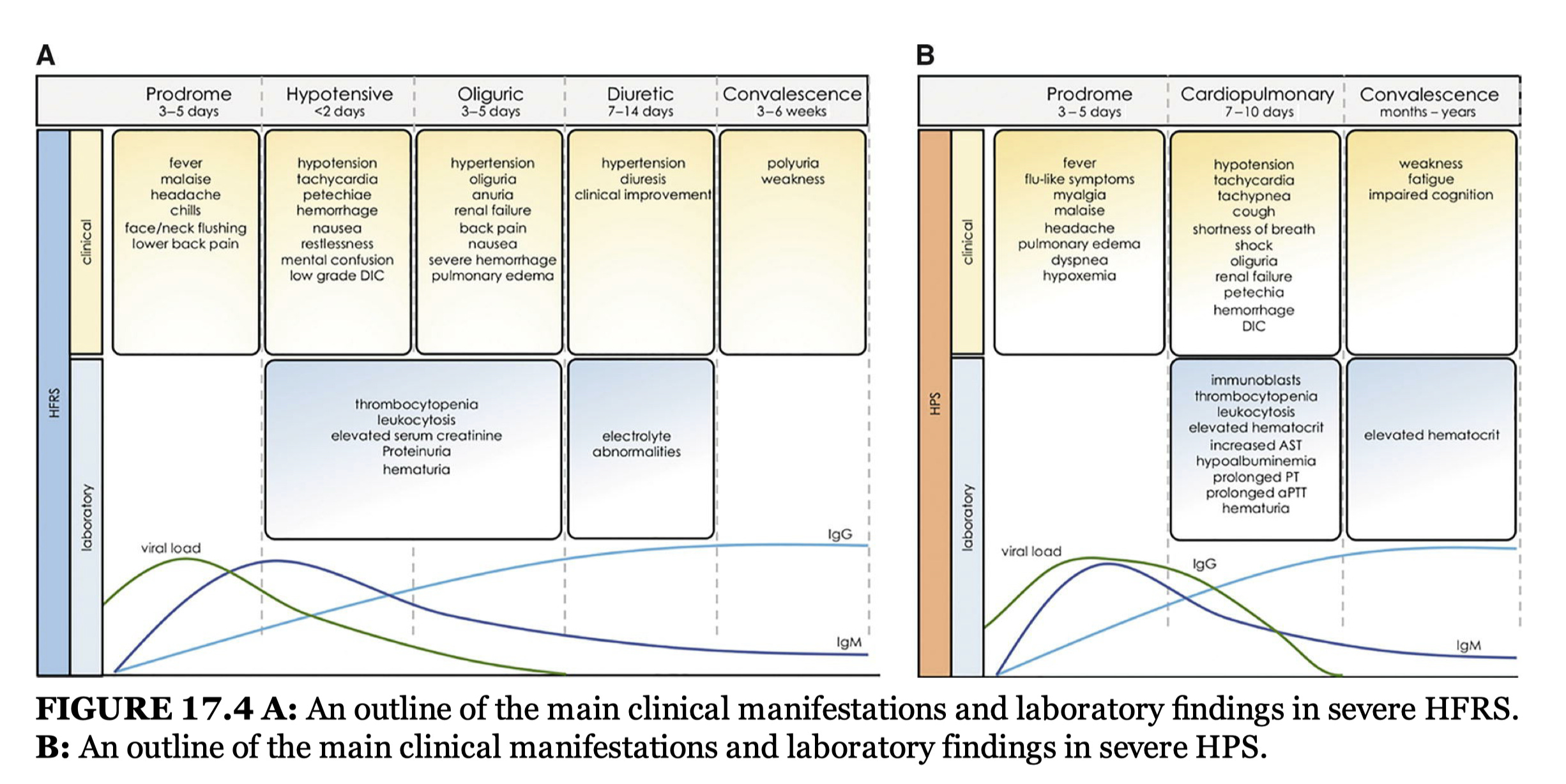
Clinically, New World hantaviruses generally preferentially affect the lungs, causing hantavirus pulmonary syndrome (HPS), aka hantavirus cardiopulmonary syndrome (HCPS), while Old World hantaviruses generally preferentially affect the kidneys, causing hantavirus hemorrhagic fever with renal syndrome (HFRS). Figure 17.4 from Fields Virology: Emerging Viruses 7th Edition below summarizes both conditions:
There is significant overlap in the two, and despite the names, pulmonary involvement may occur with HFRS and renal involvement may occur in HPS. The two diseases may reflect a continuum of one condition. Nonetheless, the phases of illness are distinct and hypertension during the oliguric phase (probably due to hypoperfusion of the kidneys triggering activation of the renin/angiotensin/aldosterone response) in HFRS clearly differentiates the two.
In this case, the leading candidate for the cause of the outbreak is a hantavirus known as Andes virus, which makes the situation more complicated. In general, hantaviruses, fortunately, do not spread from person-to-person. While it does not appear to accomplish this easily in the way that, for example, SARS-CoV-2, influenza, or measles do, Andes virus has demonstrated the capacity to be spread from person to person. A 2021 meta-analysis did conclude that there was not enough evidence to claim that Andes virus is capable of person-to-person transmission; however it did not include the previously linked 2018-2019 outbreak in Chubut Province, Argentina in its analysis. In that particular outbreak, the timing of cases and genomic epidemiology make it very difficult to argue that person-to-person transmission did not occur given that the timing between the onset of symptoms between cases fits well with the incubation period of the virus and sequencing confirms a common source. Notably, spread of the virus was driven by a few superspreaders. It appears that transmission may occur when infected individuals are in contact with uninfected individuals over close distances, for prolonged periods, while symptomatic in the early stage of illness such as the prodrome or the beginning of the second phase of HPS or HFRS (at least, those are the conditions in which transmission appears to have occurred in the outbreak in Chubut Province). Sexual contact is associated with a much greater rate of transmission than casual contact (17.6% vs 1.2%) and viral RNA can be found in semen for years after infection; that said, it does not prove that Andes virus is sexually transmissible in humans per se (i.e., infects via exposure to the semen or vaginal fluids of infected patients), as viral RNA is not the same as replication-competent virus capable of causing disease. Clear evidence of transmission from an asymptomatic source is not available, though virus RNA can be detected in the blood up to 2 weeks before symptom onset. Nonetheless, such conditions can be readily attained on a cruise ship. Additionally, Andes virus has a particularly high case fatality ratio associated with it among hantaviruses, commonly stated to be around 40%.
Treatments and Vaccines?
There are presently no specific treatments for hantavirus infections available. Some inactivated vaccines in Asia (specifically Korea and China) targeting native hantaviruses (Hantaan virus and Seoul virus) do exist, but their effectiveness is unclear (in large part because these are rare infections). Management of hantavirus cardiopulmonary syndrome is centered around supportive care including measures like maintaining adequate blood pressure to perfuse the organs, supplemental oxygen, invasive mechanical ventilation, and extracorporeal membrane oxygenation. Hemofiltration may be needed to manage fluid overload. With Andes virus specifically, hemorrhage is common, but this is not the case for hantaviruses broadly. Early initiation of high acuity supportive care associates with better outcomes.
Ribavirin has shown effectiveness in Hantaan virus infection manifesting as hemorrhagic fever with renal syndrome with a sevenfold reduction in mortality, but it is not clear how well it would work for other hantaviruses. Anemia predictably occurred from the use of ribavirin but reversed upon discontinuation of the drug. Some evidence suggests that ribavirin also has activity against Sin Nombre virus. Nonetheless, an open-label study of ribavirin failed to demonstrate a benefit for the drug against Sin Nombre virus, though limitations given the study design apply.
Use of convalescent plasma has been associated with reduced mortality against Andes virus, but there is significant uncertainty in the extent of the effect and conclusions are limited by the nonrandomized nature of the study. Similarly, data suggests that hantaviruses including Andes virus use β3 integrin (CD61) as a receptor to enter cells (generally in complex as αIIbβ3 and αvβ3), and so antibodies (or their antigen-binding fragments) targeting β3 integrin to prevent engagement with Gn/Gc have been put forth as possible therapeutics. Newer work, however, finds protocadherin-1 is a key receptor for New World hantaviruses. This is expressed on lung endothelial cells, but not renal, and cannot be used for entry by Old World hantaviruses. This potentially explains the difference in organ tropism between the two groups. Antibody against protocadherin-1 was able to prevent entry of New World hantaviruses in pulmonary endothelial cells.
Given the huge diversity of hantaviruses, pursuit of a vaccine to cover all of them (or many of them) is difficult. The nucleoprotein of hantaviruses is relatively well conserved (particularly the groove that binds RNA), but its use as a conventional vaccine antigen is not immediately intuitive as it is internal to the virus and thus should not be accessible to antibodies (though infected cells would be susceptible to killing by T cells—which may be a viable path to protection). There may be another mechanism by which antibodies against nucleoprotein might be protective, however. Antibodies against the nucleoprotein of crimean congo hemorrhagic fever virus, a bunyavirus like hantaviruses, have been shown to induce robust protection in a rodent model through their interaction with TRIM21, which targets the protein for degradation by the proteasome and promotes adaptive immunity via enhanced antigen presentation. A recent preprint furthermore describes localization of hantavirus N proteins at the cell surface, as well as their transfer between cells and secretion. This suggests that antibodies against N may recognize it on the surface of cells and exert protection accordingly (i.e., through antibody-dependent cellular cytotoxicity or complement-dependent cellular cytotoxicity), as has been suggested to be the case for other viruses. Nevertheless, prior work suggests that N antibodies alone may not be enough for protection even if N-based vaccines do appear to generate at least partial protection.
Better outcomes from hantavirus infection associate with neutralizing antibodies targeting Gn/Gc, which is consistent with expectations from first principles of viral immunology. The antibodies appear to be durable among survivors. However, Gn/Gc vary substantially between hantaviruses. Still, an antibody capable of neutralizing both New and Old World hantaviruses has been identified, suggesting that a pan-hantavirus vaccine targeting Gn/Gc may be possible. Multiple broad and potently neutralizing antibodies have been isolated from survivors of hantavirus infections and these demonstrate protection in animal models. Because the incubation period for hantavirus infection can last for 1-8 weeks after the initial infection, it is conceivable that infections can be controlled before causing disease through a memory B cell response rather than requiring constitutively high levels of neutralizing antibodies, which, if true, could make the attainment of an effective vaccine easier.
Now what?
At this point, we still have more questions than answers. Key immediate goals for the investigation include establishing the mode of transmission of the hantavirus on the MV Hondius, identifying the specific type of hantavirus, quarantining exposed individuals and performing contact tracing, and treating and isolating symptomatically infected individuals. The nature of spread with hantaviruses, including Andes virus, suggests that quarantine and isolation measures should be very effective in preventing further spread, though implementability of those measures may not be trivial. The WHO is coordinating with the governments of South Africa, Senegal, Spain, and the Netherlands to address this outbreak. For now, we have to trust those brilliant professionals to do the jobs they are so excellent at and hope that the passengers and crew on MV Hondius make it through this experience as unscathed as possible.



Thanks a lot for this excellent summary
Thank you!