People are panicking over how hantavirus spreads. They seem to be missing a few key points.
Airborne or not airborne isn't the whole story.
Sentiment regarding the Andes virus (ANDV) outbreak on the M/V Hondius cruise ship has, unsurprisingly, become extremely polarized. There’s a camp of people who seem to think that public health agencies are salivating at the prospect of enacting lockdowns (whatever that means), quarantines, and vaccinating everyone (with a vaccine that does not presently exist). Another camp of people argues that the WHO and other public health agencies are being far too laissez-faire with respect to transmission of ANDV, and we must respond with airborne precautions (more on what that means in a bit) to deal with the outbreak, and anything less than that is consigning us to a pandemic of hantavirus pulmonary syndrome (HPS) that’s fatal in ~40% of cases. Crowded out by these voices are the voices of actual experts in the subject of hantaviruses and the public health and medical professionals actually responding to the situation in real time. Let’s take a closer look at what the data show.
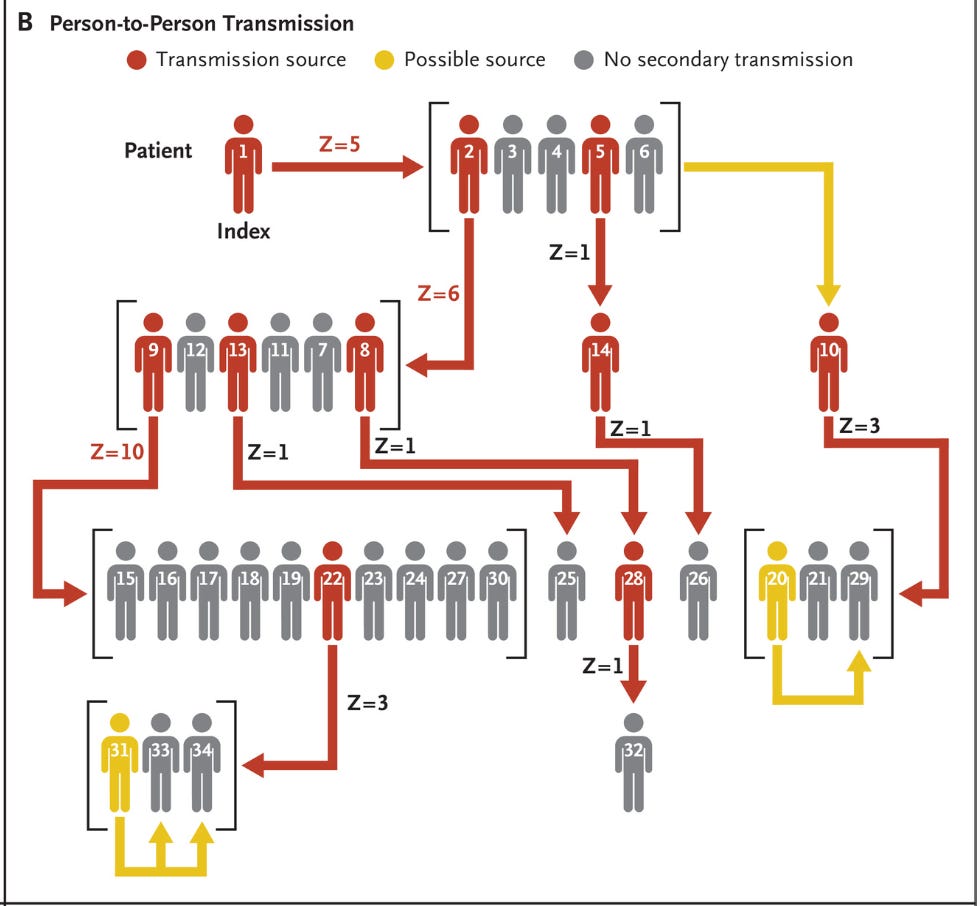
It makes sense to start with the largest outbreak of Andes virus on record, which occurred in Epuyen, Chubut Province, Argentina in 2018. The report here has some complex nuances. Let’s take it piece-by-piece. First, here’s a reconstruction of the transmission chain:
In essence, we have 4 generations of infections in the chain of transmission before the outbreak stops, and it’s driven by a relatively small fraction of people (i.e., superspreading). To clarify what the research team infers happened here: patient 1 (the original case) attended a birthday party for 90 minutes while symptomatic with a flu-like illness (fever and malaise), wherein he infected patients 2-6. Patient 2 had an “active social life” and went on to infect patients 9, 12, 13, 11, 7, and 8. Patient 2 subsequently died from their infection, and patients 15, 16, 17, 18, 19, 22, 23, 24, 27, and 30 attended the wake where Patient 9 (his wife) was actively symptomatic and they all got infected. The remaining 12 patients were each in contact with at least one previously symptomatic patient. Four patients (Patients 10, 29, 33, and 34) may have been infected by more than one person.
It’s important to note that this is a dead-end infection for the majority of these patients. Moreover, there is a link to a symptomatic patient to explain each of the cases. I’ll return to that point a bit later when examining asymptomatic and presymptomatic transmission. So the question is: how did these people get infected? That’s where things get messy.
With hantaviruses broadly, the evidence suggests that human infection from reservoir species occurs when humans inhale the virus from their urine, feces, or saliva. It would therefore make sense (before we consider the epidemiology of outbreaks for the moment) that spread is driven by inhalation between people as well, and this is supported by the fact that ANDV causes hantavirus [cardio]pulmonary syndrome—an infection of the lungs (in part) as well as the nature of contact between patients in this outbreak. Previous work has also suggested the possibility of sexual transmission given that transmission was much more likely to occur between sexual partners. However, unless you’re, let’s call it, “creative,”1 anything that spreads through the air is going to spread quite readily through sexual contact (depending on exposure time), so whether or not this is sexual transmission per se (i.e., the pathogen is present in the bodily fluids or tissues in contact with one another during sex acts and can transfer between individuals engaged in sex acts to cause productive infections) is difficult to parse. It is also potentially possible that hantaviruses may cause infections through contaminated surfaces (fomites) and this is emphasized in some guidelines regarding infection prevention, though it has also been found that compared with other hantaviruses, Andes virus is not as stable as other enveloped viruses (half-life is about 1 day). Still, that does suggest meaningful retained infectious potential over brief periods.
There is more to this particular outbreak and paper that I want to discuss, but first I have to orient you to the topic of airborne transmission.
Intermission: Airborne—what does it really mean?
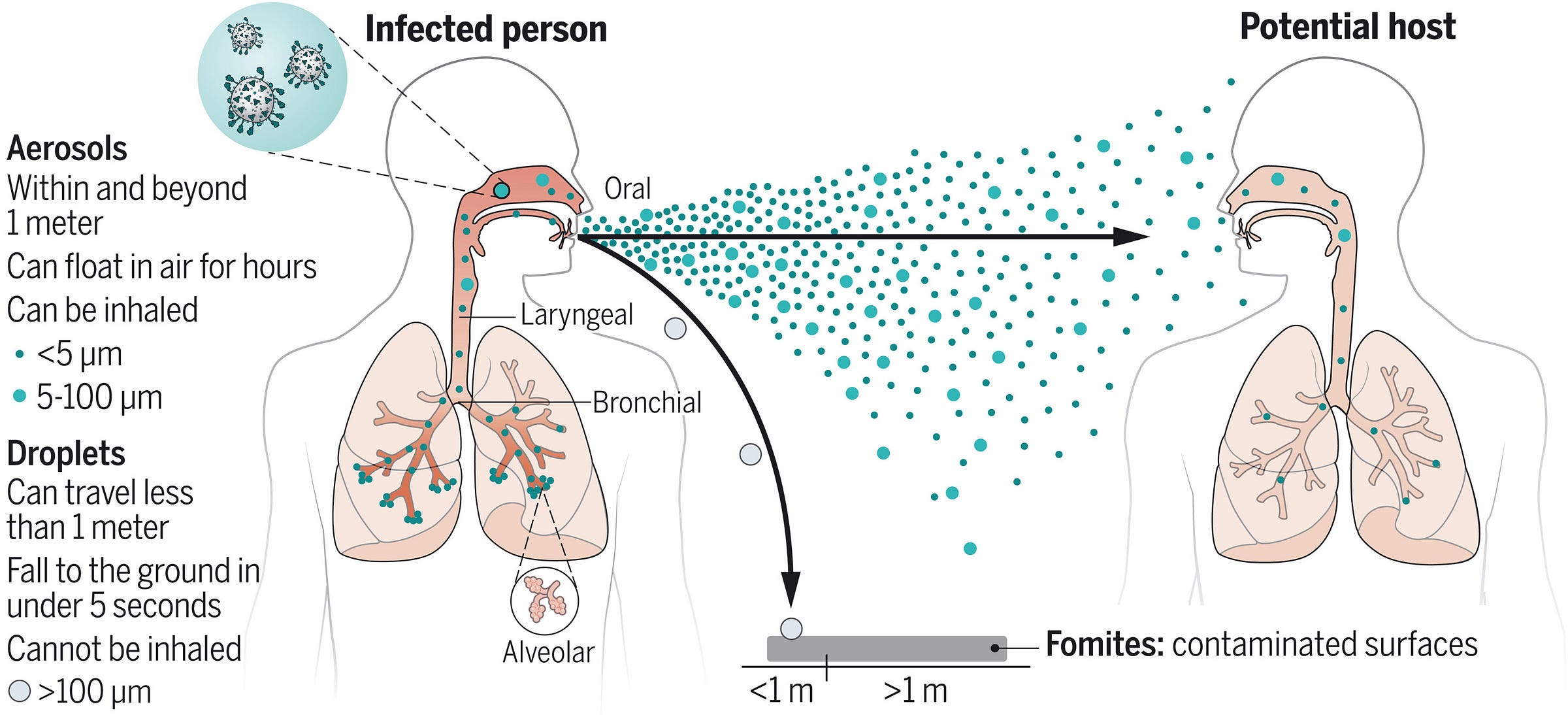
Despite everything that happened throughout the COVID-19 pandemic, the term “airborne” still seems to be a point of confusion based on what I’m seeing on social media.
Across 3 groups of people, the same exact terms can mean something quite different. To avoid confusion, I have specified previously that Andes virus is thought to be contracted by the inhalation route (I was hoping to avoid getting into this matter), but it seems that I can’t avoid elaborating if my goal is clarity.
When clinicians use the term airborne, they often are not describing a route of transmission per se, but rather specific precautions that apply for preventing transmission. In general, these precautions fall into 3 basic types. If you’ll pardon the coarseness of my summaries, the precautions are:
Contact: wearing gloves, gowns, using disposable equipment, handwashing.
Droplet: source control (e.g., putting a mask on the patient), surgical mask and eye protection, placing the patient in a single room if possible and limiting transport.
Airborne: droplet precautions but using a fit-tested respirator (such as an N95, N100, or FFP2) and isolation in a negative-pressure room/airborne infection isolation room (AIIR). Limit trafficking of people in and out of the room.
Both airborne and droplet precautions are applied to transmission routes that most people would commonly describe as airborne. The distinction is that it was long thought (largely but not completely owing to the errors of Alexander Langmuir and several others) that droplets rapidly fell to the ground at short distances (i.e., within 2 meters) because of their mass; thus transmission over long ranges could not occur. In terms of airborne transmission, it was believed that if transmission over long distances was possible (or, more properly, significant to the epidemiology of the infectious disease), the pathogen exhibited “airborne” spread, with infectious particles being small enough to remain suspended in the air for prolonged periods. It was similarly thought that simple surgical masks would not meaningfully reduce transmission of pathogens exhibiting airborne spread, which is not necessarily the case (but source control is always going to be their key role and well-fitted respirators are indeed better). With the benefit of hindsight, this does seem a bit silly. After all, it’s always going to be the case that the closer you are to the source of an infection, the greater your risk is going to be. Still, with some infections, the effect of aerosols that can transmit over long distances is quite stark. For example, it is possible to contract measles by walking into a room that someone who has measles has been in in the last 2 hours even if they are gone.
In the modern understanding: any transmissible pathogen that gets into the air and can be inhaled to cause infection represents airborne transmission regardless of the size of the fluid particles housing it, meaning transmission over long as well as short distances exists along a continuum (depending on viability, emission rate, infectious dose, ventilation, humidity, UV, deposition, and dilution). Starting from this point, there is a common and important fallacy people commit: the assumption that something capable of airborne spread must have a massive R0 value (i.e., in a population lacking immunity, each case can generate a huge number of secondary cases). I think Tang et al. in their 2021 paper put it well here (emphasis mine):
Myth 4: ‘if the basic reproductive number, R0, is not as large as for measles, then it cannot be airborne’
The basic reproductive number (R0) is generally defined as the average number of secondary cases arising from the presence of one single infected ‘index’ case in a population of uniformly distributed but otherwise totally susceptible individuals. The key problem with this statement is that R0 is not related directly to whether or not a disease is transmitted through aerosol inhalation. R0 signifies how many people become infected after contact with one infected person, but the mechanism of transmission is irrelevant. Various organisms can be disseminated by the airborne route but are not necessarily transmitted person-to-person. For example, hantaviruses, which cause hantavirus pulmonary syndrome, and Bacillus anthracis, which causes anthrax, have animal reservoirs and are acquired by inhalation, but they are not transmitted person-to-person. They have R0 = 0 yet they are considered to be airborne diseases [43,44]
So, basically, airborne does not automatically equate to transmissible, regardless of the distance. Still, it’s not as though distance does not matter. For example, as you get farther from the source, the particles in the air progressively become less concentrated and house fewer infectious particles. It is therefore possible to get to a distance where a minimal infectious dose cannot be acquired or at least is much harder to acquire.
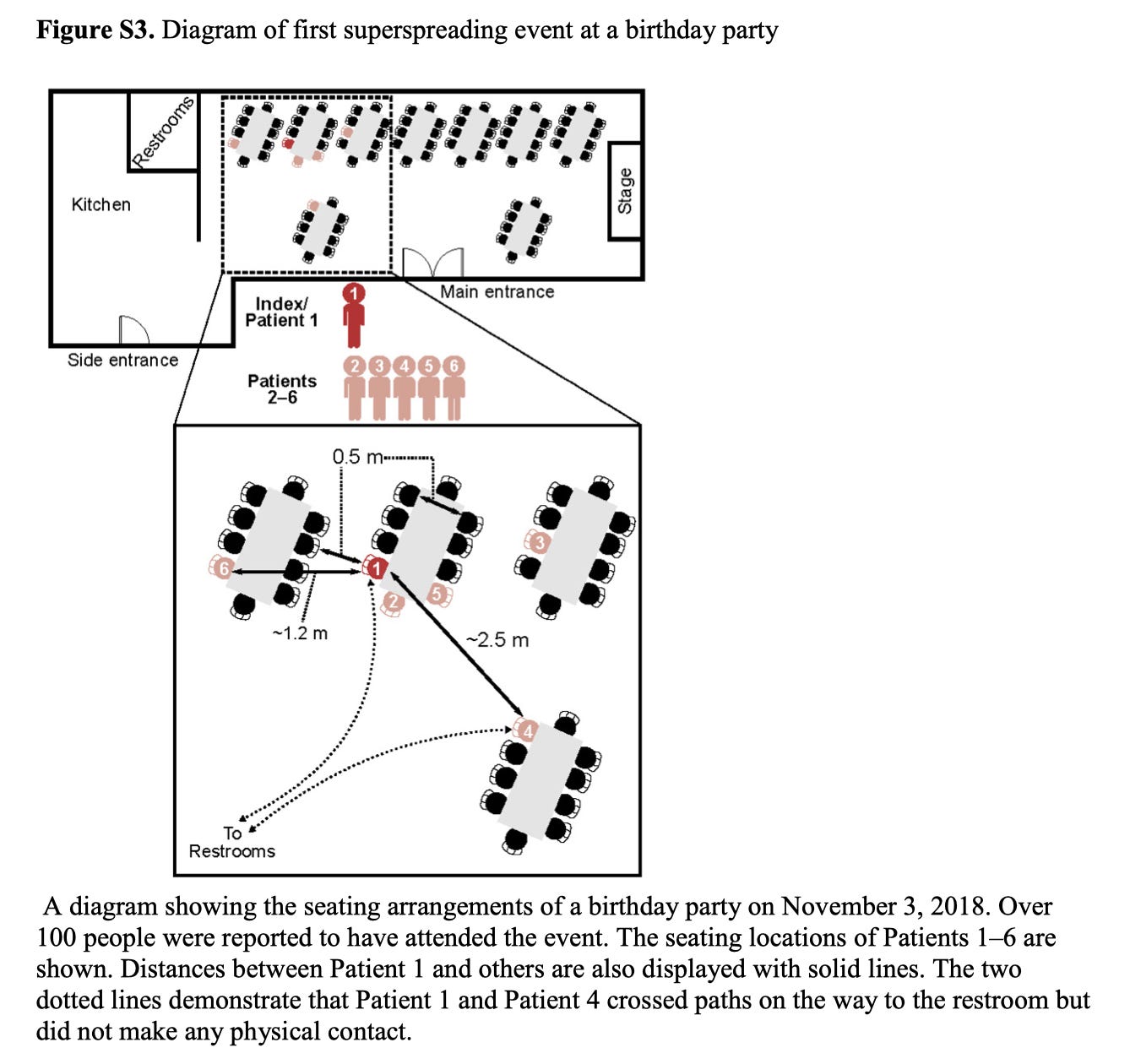
Let’s now return to the 2018 outbreak data. While not discussed in the main body of the text, the supplementary appendix has some interesting notes:
Patient 4 was seated farther away from patient 1 (i.e., beyond what we would expect for “droplets” to cause infection) and it is reported that they only got close to each other briefly while passing on the way to the restroom without physical contact. Table S7 contains additional notes about inferred transmission events.
And now, here is where things become very tricky, because, I am afraid, we now need to know not just about the nature of pathogen transmission but about the limitations of outbreak investigation. Per the paper:
Data regarding epidemiologic factors and the close contacts of infected persons were retrospectively obtained by means of patient interviews at the clinic and at follow-up visits to patient homes by public health authorities throughout the study.
There is nothing fundamentally wrong with this method. It is the cornerstone of outbreak investigation. However, it cannot be ignored that there are recall biases arising from this approach and information obtained in these investigations is rarely complete. A systematic review from 2021 by Toledo et al examining the transmissibility of ANDV between people ultimately concluded that the available evidence had significant biases that did not allow for the conclusion that ANDV is transmissible between people (note that this is not the same thing as saying that ANDV does not spread between people—it means that the evidence quality is too poor to conclude that). It states the following in regard to the 2018 outbreak (emphasis mine):
No investigation of possible co-exposure to the excreta/secreta of infected rodents in the environment seems to have been conducted and the possibility of environmental exposure is not even considered as a possible limitation of the study.
…
While the use of genetic sequencing data to confirm human-to-human transmission has been used in investigations of clusters in Argentina and Chile [7–11, 25, 26, 32, 35], it has the limitation of only providing strong evidence when cases that occur in different geographic areas have identical sequences. “When all case-patients remain in the disease endemic area … molecular dissimilarities rule out person-to-person transmission, but identical sequences do not support it” [10].
To complicate things further, failing to identify a rodent source in the beginning of hantavirus outbreaks does not rule out that one is driving the outbreak, as was seen in the 2013 outbreak of Sin Nombre Virus in Yosemite. There is also the fact that there is a huge span in the incubation period of ANDV and substantial variability between individuals individual incubation periods means that definitively identifying a transmission event is exceedingly difficult. Having said that, Toledo et al’s conclusion (“The balance of the evidence does not support the claim of human-to-human transmission of ANDV.”) is more extreme than I am comfortable with under the circumstances and the precautionary principle suggests we should not assume this to be the case (especially when the risk is a devastating infectious disease with a ~40% mortality ratio).
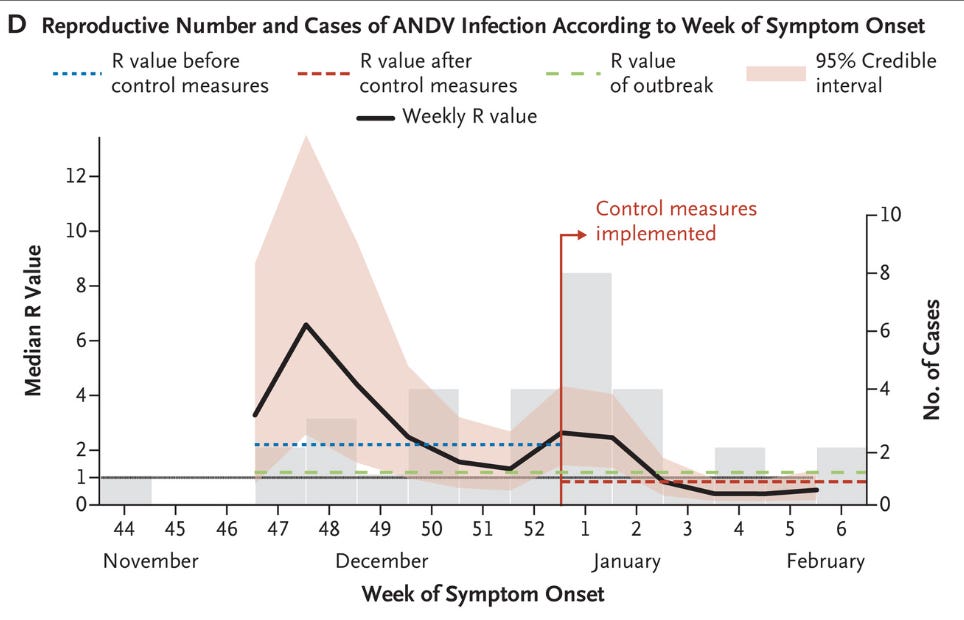
Still, this means that the cases in Table S7 may reflect long-range aerosol transmission, or, may be the victim of biases inherent to this type of study that actually reflects transmission over shorter distance. It’s very difficult to know. Should we rule out the possibility of long-range airborne transmission? I think that would be a mistake. But that’s a separate (though related) question from what precautions should be taken in this outbreak. Returning to the 2018 outbreak, here is the trend in the R value (the effective reproductive number) during the course of the outbreak.
Sustaining transmission requires maintaining R above 1 (a value of 1 means that each infection generates, on average, one infection to replace it; a value above 1 means that each case generates, on average, more than one case, meaning the outbreak gets bigger). Once control measures were implemented, note what happens to R: it drops rapidly below one (i.e., each case generates an average of fewer than 1 case to replace it, meaning the outbreak size progressively decreases). What were the control measures taken?
1.8 Further details on case isolation and enforced self-quarantine measures
Provincial and national public health authorities coordinated the establishment of isolation for confirmed ANDV cases and self-quarantine for high-risk contacts. High-risk case-contacts were identified as those who were in close contact for more than 30 min with a confirmed and symptomatic ANDV case. Case-contact self-quarantine was imposed around December 31, 2018 (i.e., after identification of Patient 18). In addition, massive social gatherings were prohibited in the affected area. Suspected high-risk contacts were recommended to remain at home and self-quarantine for at least 40 days and were asked to wear N95 respirators in the presence of others. Control measures were lifted in March 2019.
Essentially, the bread-and-butter infectious disease epidemiology measures. But there’s another subtlety here too. It’s easy to focus on who got infected in this outbreak, but who didn’t also matters a lot:
Although several high-risk medical procedures were performed in patients with ANDV hantavirus pulmonary syndrome, including orotracheal intubation and cleaning of bodily fluids such as vomit, diarrhea, and other secretions, no nosocomial infections were reported among health care workers who had been in direct or close contact with the patients at the health care facilities (Hospital Esquel Zonal and Epuyén Rural Hospital). Approximately 82 health care workers were exposed to symptomatic patients with confirmed ANDV infection at Hospital Esquel Zonal from December 2 to December 13, 2018. Of the 45 persons who worked in the intensive care unit and emergency department, only a small number used any form of personal protective equipment (including N95 respirators [N100 respirators for intubations and cleaning], goggles, and disposable laboratory coats) while they were in direct contact with an infected patient.
This was also seen in the US in January 2018, wherein a woman who visited the Andes region of Argentina and Chile contracted ANDV and returned to the US. What happened?
Among 53 contacts identified in six states, 51 were successfully contacted (Table). Of these, 28 were health care personnel, 15 were airline contacts (flight crew who served the traveler and passengers seated within one seat of the traveler), and eight were other contacts of the traveler (including acquaintances and a hospital roommate). All contacts were advised to self-monitor their temperature daily for 42 days from last contact and to seek medical evaluation for any of the specified symptoms. Contacts who developed symptoms were tested for hantavirus by RT-PCR and serology by CDC’s Viral Special Pathogens Branch.
Two high-risk contacts were identified: a health care worker with exposure to the traveler’s sweat and a family member with exposure to the traveler’s clothes and bedding. Both high-risk contacts remained asymptomatic. Six low-risk contacts, all flight attendants, reported influenza-like illness, diarrhea, or mild rhinitis during the incubation period; all tested negative for hantavirus by RT-PCR and serology. The remaining low-risk contacts remained asymptomatic, and the investigation concluded on March 8.
Not one of her 51 contacts seemed to get ANDV.
There is also the question of transmission from asymptomatic individuals. There are few cases of ANDV infection documented and even fewer that are believed to be the result of human-to-human transmission, which means that a precisely defined infectious period is not really possible with the available data. Nonetheless, if asymptomatic transmission were important to the epidemiology of the disease, we would expect to see cases that we could not trace to the outbreaks or contact with patients exhibiting symptoms. HPS is not a subtle infectious disease. While not dispositive, the fact that putative cases of HPS that have not been connected to exposure to an asymptomatic individual suggests this is probably not a major contributor to the epidemiology of the disease.
The question of presymptomatic transmission is more complicated. It could be argued that the detection of cases linked to exposure to a symptomatic individual reflect an ascertainment bias, and do not rule out the possibility of transmission before symptoms occur. There are serosurveys suggesting that we may miss mild infections by ANDV and other hantaviruses.2 That said, at the level of public health, the concern is less “how ANDV is spread?” in the broad sense, but rather “how does ANDV spread to result in the devastating clinical entity that is HPS?” In a statement by the International Hantavirus Society they write:
The precise timing of infectiousness remains incompletely defined. While symptomatic patients are likely to represent the highest-risk group, available outbreak reconstructions do not support overly categorical statements that transmission can occur only after clear symptom onset. Transmission potential during prodromal, early symptomatic or minimally symptomatic phases, should be considered when designing contact tracing, testing and quarantine strategies.
Nonetheless, while we are constrained again by the limited number of cases, this would be most readily supported by a transmission event through contact between someone infected before they displayed symptoms (most transmission events reported seemed to occur during the prodrome i.e., flu-like illness preceding full-blown HPS). So far, we haven’t seen that. That does not rule it out, but I would argue it downgrades the epidemiological importance of those exposure as means of transmission.
What about virus RNA?
I refer you here. Virus RNA is not the same thing as infectious virus and we must be cautious about inferring infectiousness from the presence of RNA alone. That does not mean that the detection of RNA should result in a default presumption of “uninfectious until infectious particles can be confirmed,” and thankfully, it doesn’t seem that anyone is doing that.
So, how should we describe transmission of ANDV?
I think that simply describing the route of transmission is not complete. If I had to summarize:
The evidence is strongest that ANDV person-to-person spread of ANDV through close, prolonged contact with a symptomatic individual (particularly during the prodrome of their illness) seemingly through the inhalation route. It therefore makes sense to prioritize infection prevention and control measures targeting this route. The extent to which people who do not develop symptoms or people who are in the phase of illness preceding the onset of symptoms is unclear but should not be excluded as possible.
The possibility of transmission over longer distances or during brief periods of contact is not disproven, but evidence for it is not as robust as for short distances and must be considered with attention to relevant limitations inherent to outbreak investigation. Nonetheless, it should be considered as possible as part of outbreak response.
Transmission through infectious surfaces or sexual transmission via contact with bodily fluids or tissues is considered a possibility, but lacks robust supportive evidence owing to confounding with other routes of exposure.
ANDV remains a virus that does not readily infect humans regardless of exposure scenario. Sequences obtained from isolates in this outbreak demonstrate a high degree of genetic similarity to the isolates taken from the 1997 and 2018 outbreaks and minimal variation across patients. That suggests that it is unlikely that this virus’s behavior will be substantially different from that of previous outbreaks (which were highly responsive to bread-and-butter outbreak control measures).
Collectively, this suggests the risk of infection in this outbreak for anyone not coming from the M/V Hondius is very, very low. The totality of evidence firmly supports that this outbreak lacks the potential to develop into a pandemic. The average person does not need to spend energy tracking everything happening unless they are personally interested.
It’s important to get this right in the general sense because we need to control this outbreak, but it is important to get it right in the narrower sense so that we know who counts as a contact, who to prioritize for testing, and to ensure everyone who needs to be monitored is monitored. We are going to hear a lot about suspected cases over the coming weeks. It is more important to not miss anyone who could be a case than to fail to capture someone who could be, which means that many of these cases will end up being false alarms. That’s supposed to happen.
Additional infection prevention precautions beyond baseline measures are not warranted for the general public and shifting to greater caution because of the risk of ANDV is somewhat misaligned with respect to infectious disease risks (for example, summer is approaching, and that means we should expect a wave of SARS-CoV-2, a virus that still causes over 100,000 hospitalizations and over 10,000 deaths per year in the US, as well as poorly understood post-acute sequelae).
What precautions (i.e., airborne vs droplet) should be taken for passengers disembarking from the MV Hondius?
It is so far beyond my pay grade to make recommendations on that that that it may as well be floating in the Andromeda galaxy. I’m going to leave that to the WHO and other public health authorities actually responding to the outbreak. Precedent establishes that they know what they’re doing with Andes virus outbreaks.
That said, placing infected individuals in biocontainment units is about the highest, most conservative level of caution that you could offer.
I tried to locate the original document but it appears it was taken down. In any case—and I say this completely seriously—I think NYC DOH’s guidelines for safe sex during the COVID-19 pandemic represented absolutely brilliant science communication that balanced the uncertainties of the time period with avoiding being prescriptive. More than that, it was presented in such a way that the guidance got disseminated widely, making it easy for many people to learn about harm reduction methods they could apply to help avoid getting COVID. That might be because of the inherent nature of the subject rather than the guidance itself, but regardless, it did an excellent job.
While the rates of seropositivity when examining ANDV specifically (as distinct from other hantaviruses in the Americas) may seem low, it is important to be cognizant of the fact that there is significant antibody crossreactivity between ANDV and Sin Nombre Virus (SNV) nucleoproteins, and thus looking at hantavirus seropositivity generally in a place where ANDV is endemic is likely a reasonable approximation for the seroprevalence of ANDV. Animal models suggest that there is also meaningful crossreactivity between antibodies against SNV and ANDV glycoproteins (Gn/Gc).




Another great article... Thanks very much! Two thoughts:
1. It's a bit weird to see so many people of varying stripes lately getting out of their trees over what very likely is a non-pandemic, little mutating (albeit narrowly serious) disease... when, as you correctly point out at the end, many thousands of Americans are already and still dying annually from COVID (almost 5,000 thus far in 2026 alone, per the CDC's data). And that fact continues to get almost no ongoing public attention or acknowledgement.
2. When I see the notion of "self-quarantine" as a control measure for hantavirus risk cases, I get kind of queasy.... If anything, the COVID pandemic markedly increased the share of the strident folks for whom "me and my needs" are the sole consideration, and how their actions impact the surrounding community is of little to no concern. I'll be waiting to see the forthcoming photos of some self-home-quarantined risk patient out at their local pub or Olive Garden for all you can eat pasta night..... :-(